The different types of pipette tips (and when to use them)
Written by Jonathan Harkins
02. April 2021
The first thing you ask yourself in the morning, after pondering if you can hit snooze one more time, is: “What should I wear today?” Your decision is influenced by many internal and external factors, such as your mood, the weather or whom you are going to meet.
Similarly, many factors have to be considered when choosing the right pipette tips for your task. Hopefully, this time the decision is not influenced by your whims, but by relevant factors such as the property of the liquid you are pipetting, and the type of application. But how do you know which pipette tip is best to pick? Here, we discuss the importance of sterility, quality and fit.
Table of contents
1 Different types of pipette tips
Non-sterile, sterile, filtered, unfiltered, long, short, low retention, wide bore – the variety of pipette tips available can be overwhelming. So let’s explore what each of these options is good for:
1.1 Non-sterile vs. sterile
It's common sense that sterile pipette tips should be used for applications where sterility is important. But can you buy non-sterile tips and autoclave them yourself to save money? In theory, the answer is yes. However, you need to make sure that the manufacturer declares them as autoclavable, and be aware of the following:
- Lack of quality control
Tip manufacturers validate their sterilization process, and perform regular quality checks. In contrast, individual labs don't usually test the effectiveness of their autoclaving process, which can lead to contaminated samples. - Autoclaved tips aren't free of RNase and DNase
Tips you’ve autoclaved yourself are sterile, which means that they are free from living organisms, but not necessarily from RNase and DNase. If you need to perform sensitive assays where this is required, you should opt for sterile pipette tips from a manufacturer who can certify that their tips are free of RNase and DNase.
1.2 Filter tips
Every time you aspirate liquid, aerosols are generated inside the pipette tip. If you don't use filter tips, these aerosols may contaminate your pipette and, consequently, your next samples – even if you change tips in between.
For example, when performing PCR applications, the cross-contamination of samples by aerosols in the pipette could lead to false positive results, as even the smallest quantities of DNA from a previous sample could be amplified. It's particularly important to use filter pipette tips when handling liquids that could damage your pipette, such as radio-labeled or corrosive samples, for both your own safety and lifetime of your pipette.
Here's a list of liquids that should always be pipetted with filter tips, as they could either contaminate or damage your pipette:
- RNA/DNA solutions
- Infectious samples
- Radio-labeled samples
- Volatile, corrosive or viscous samples
- Strong acids or bases
And last but not least, filter pipette tips can be useful for training new lab staff. Spending extra money on filter tips until your colleagues get used to your instruments is a good idea that usually pays for itself, as you can avoid pipette contamination or damage from liquid entering the lower end of the pipette.

1.3 Long tips
Have you ever risked cross contamination by putting the shaft of your pipette into a tube for a standard tip to reach the bottom? To eliminate this risk, many manufacturers offer extended length pipette tips suitable for labware such as microcentrifuge tubes or deep well blocks.
1.4 Short tips
Short pipette tips offer two advantages. First, they support the targeting of small wells, e.g. when manually pipetting into a 384 or 1536 well plate with a multichannel pipette. And second, they offer improved ergonomics by allowing you to pipette closer to the bench, reducing the strain on your arm.
1.5 Low retention tips
As the name suggests, low retention pipette tips retain less liquid, providing more accurate and consistent results while saving precious reagents. However, they are more costly than standard tips, so you should know exactly when it is worth using them. Our in-house application scientist demonstrated, in a series of tests, that both standard and low retention tips provide ideal liquid recovery when pipetting water, but produce significantly different results when viscous or low surface tension solutions are handled.1 Therefore, low retention tips are ideal when pipetting highly concentrated, and consequently viscous, samples during:
- PCR, cloning, sequencing or other DNA and RNA applications
- SDS-PAGE, protein purification or other protein analysis applications
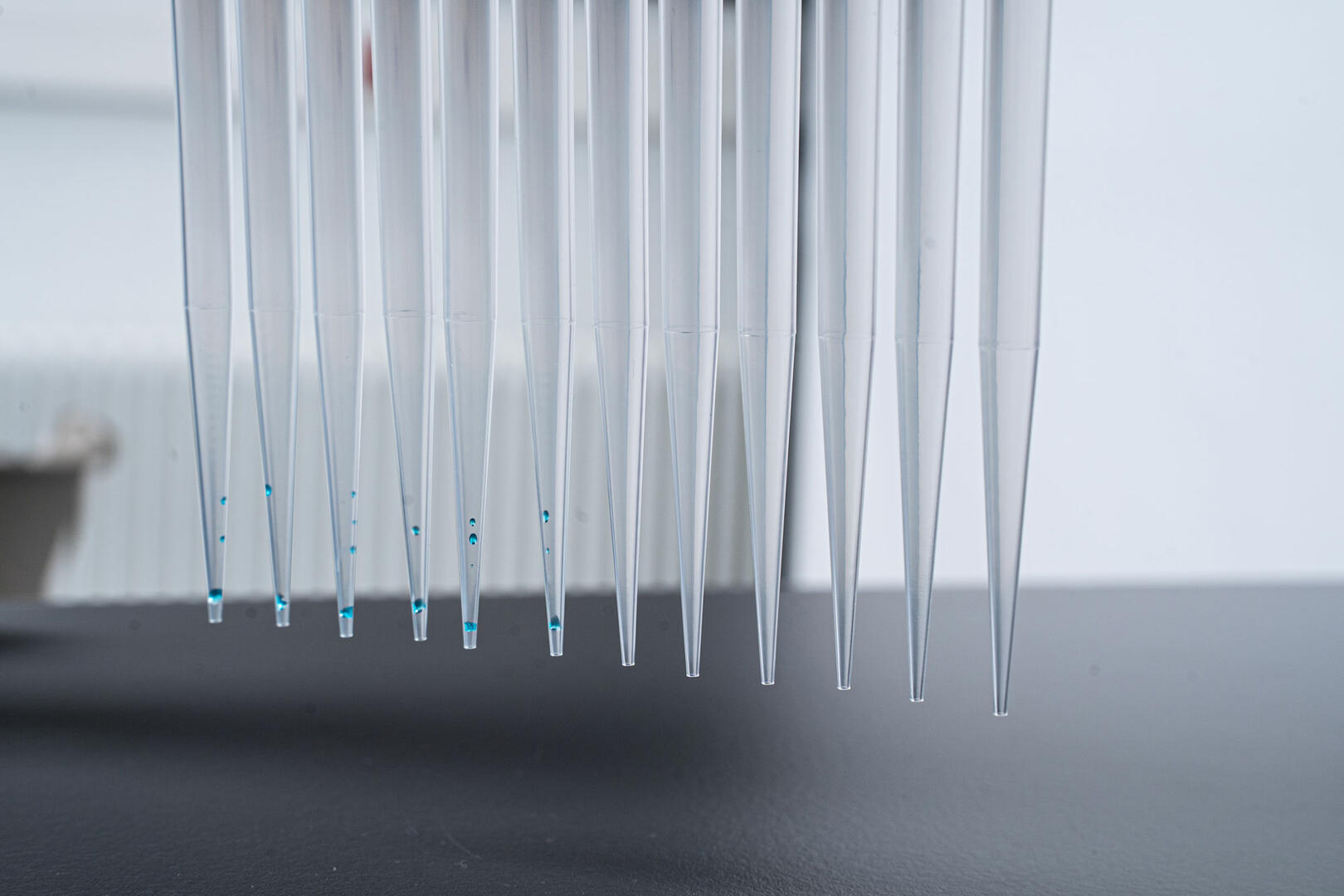
Are all low retention tips the same?
When producing low retention pipette tips, manufacturers typically use either a different polypropylene blend to their standard tips, or add a silicone coating. Both techniques prevent viscous or low surface tension liquids from spreading out and 'wetting' the inner wall of the tips, however, the latter has one major disadvantage – a silicone coating can wash or leach out with your sample. So you should always choose pipette tips with a polypropylene blend offering heightened hydrophobic properties to ensure that liquid-repellents can't contaminate your samples.
1.6 Wide bore tips
Fragile cellular samples can get damaged when they are forced through the narrow orifice of standard pipette tips. Therefore, you should use wide bore tips when transferring cellular samples, such as fragile cell lines, or other viscous materials. The wider orifice of these tips prevents (cell) shearing and reduces flow resistance.
2 Why tip quality and fit are crucial
Now that you know when to choose each type of tip, we need to address tip quality and fit. Low quality or poorly fitting pipette tips can negatively impact the reproducibility of your results. This means that you will need to repeat experiments, wasting valuable time and money. On top of that, repeating experiments means even more pipetting, which increases the strain on your arm, wrist and fingers – especially if you use poorly fitting tips that require high attachment and ejection forces – and can ultimately lead to repetitive strain injuries.
2.1 How to assess tip quality
The first factor influencing the quality of pipette tips is the polypropylene blend. High quality tips are made from virgin polypropylene, which is free from plastic and/or metal additives that could contaminate your samples. Checking that a manufacturer doesn't use metal additives is especially important when buying colored tips, as metal additives can often be found in dyes.
The second factor influencing quality is the injection molding machine. The slightest batch-to-batch or within batch variations – such as differences in straightness, molding flash or streaking – negatively impact the accuracy and precision of your results. These irregularities often can't be seen by the naked eye, so it is better to avoid buying the cheapest tips on the market, to reduce the risk of inaccurate and imprecise results.
2.2 How to find a properly fitting tip
Tip shapes are not universal, so not every tip will fit your pipette. Ideally, you should only use the tips recommended by your pipette manufacturer. If you use tips that haven't been tested and validated by your manufacturer, you should always gravimetrically test their performance to ensure that they produce reliable results.
3 Conclusion
Buying high quality, properly fitting pipette tips might seem more expensive at first glance, but will save you a lot of time, money and health issues. However, the question of whether you can work with non-sterile standard tips, or whether you need to invest in tips with features such as heightened hydrophobic properties or wider orifices, depends on your requirements. Make sure to answer this question every time you start a new application by following our hints above, so that your pipette tips will never compromise your lab work again.
Do you have an additional tip on tips you want to share with the community? Post it in the comment section below!
Further reading: