Comparison of RNA and DNA extraction methods
Written by Éva Mészáros
27. May 2021
Nucleic acid extraction (NAE) is the initial step for many molecular biology applications, so one of the first questions you have to answer when setting up a lab is what kind of RNA and DNA extraction method to use. We've recently assessed different options when setting up an in-house lab, and compiled our evaluation to help you shortcut the process. This blog post represents the second part of a series of five articles on how to get from samples to pure nucleic acids.
Table of contents
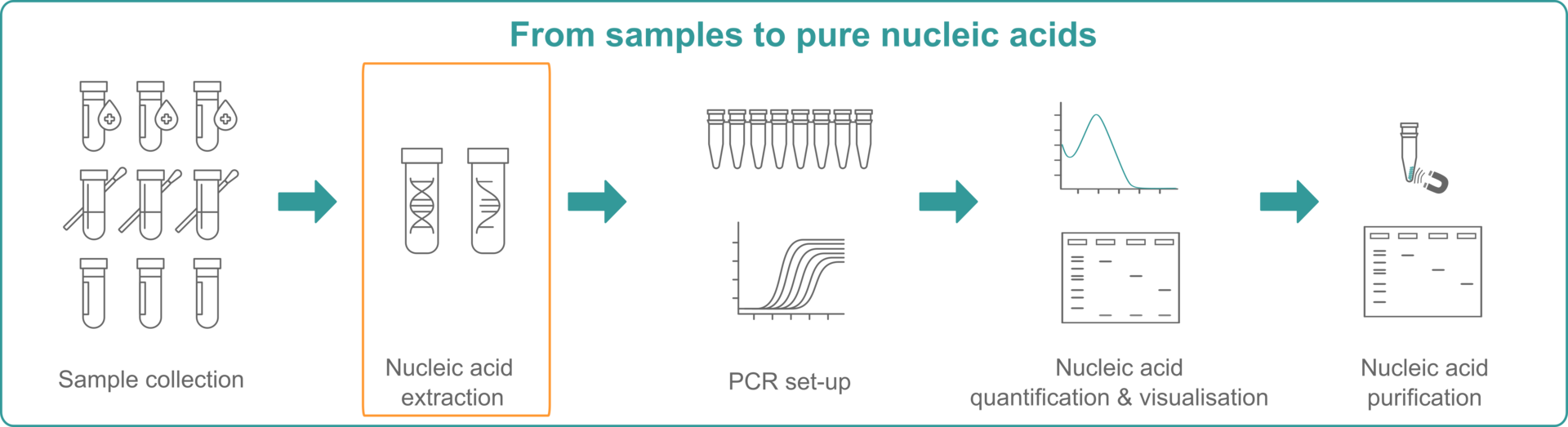
What is nucleic acid extraction?
The first nucleic acid extraction was performed in 1869 by the Swiss physician Friedrich Miescher, who isolated DNA from leukocytes when he was trying to determine the chemical composition of cells.1 Since then, nucleic acid extraction has been extensively studied and further developed to be defined as follows:
Nucleic acid extraction
To extract DNA, RNA or total nucleic acids from samples – such as blood, cultured cells, microbes, soil, or plant and animal tissues – the samples undergo a varying set of manipulations, depending on the sample and downstream application. These include cell lysis, inactivation of nucleases, and purification to separate the desired nucleic acid from cellular debris.
RNA and DNA extraction methods
To extract a sufficient yield of high quality, purified RNA or DNA for your downstream applications, you have to find the best extraction method (or combination of methods) for your sample type. This chapter gives an overview of the most common methods, explains how they work, and discusses their advantages and disadvantages. We’ll also tell you what kind of equipment you need for which method, and what we work with in our own lab.
But first, let's take a look back at two nucleic acid extraction methods that have been developed over the last 150 years, and show you why they should no longer be used, even though they are quick and easy to perform.
Conventional methods
Rapid one-step extraction
As the name implies, this method consists of only one step. An extraction buffer containing Tris-HCl, EDTA, sodium lauryl sarkosyl and water insoluble PVPP is added to the sample. The mixture is then incubated, cooled and diluted in double-distilled water.2 Sounds great but, unfortunately, this method lacks any purification steps, so often leads to inaccurate or unreliable results. Although you would be able to extract nucleic acids from non-complex samples – such as Gram-positive bacteria – with this method, it is no longer used because PCR inhibitors are co-extracted, and buffer substances (e.g. EDTA) are carried over and can strongly influence downstream applications. On top of that, today's applications – such as qPCR or next generation sequencing (NGS) – are very sensitive, and require nucleic acids with high purity.
Chelex 100 extraction
For Chelex 100 extraction, an extraction buffer containing Chelex 100 resin, SDS, NP40 and Tween® 20 is added to the sample. The mixture is incubated at 100 °C for 30 min, then centrifuged. The supernatant is removed, and adjusted to a final concentration of 10 mM Tris-HCl and 1 mM EDTA.2 Due to a lack of purification steps, this method also results in low purity, and cannot efficiently remove PCR inhibitors from complex matrices. In addition, the high temperature and alkalinity of the protocol can denature the DNA.3 Therefore, Chelex 100 extraction is rarely used today when alternative methods, such as spin column extraction, take the same amount of time and offer much better results.
Furthermore, conventional RNA and DNA extraction methods are not suitable for tough-to-lyse samples (e.g. yeast, human specimen, animal tissue or plant material), which require both chemical and physical cell lysis steps.
Most common methods
Most labs now isolate nucleic acids by using sample- and application-specific kits for spin column or magnetic bead extraction workflows. But before we look at these methods in detail, we'll briefly discuss some solution-based methods. These can be useful for sample types that don't provide the desired output with spin column or magnetic bead extraction, e.g. samples with large particles such as soil or dust, or high volume samples such as chemostat cultures.
Solution-based methods
CsCl density gradient centrifugation with EthBr
To extract nucleic acids using cesium chloride (CsCl) density gradient centrifugation with ethidium bromide (EthBr), you need to mix your lysed samples with CsCl and EthBr and subject them to high speed centrifugation. CsCl is an extremely dense salt, and will set up a concentration gradient, whereas EthBr will intercalate into nucleic acids, which separate into bands according to their density. Since EthBr becomes fluorescent under UV light, you can easily locate and extract the nucleic acid bands. EthBr can subsequently be removed from the extracted nucleic acids using ethanol precipitation.3,4,5,6
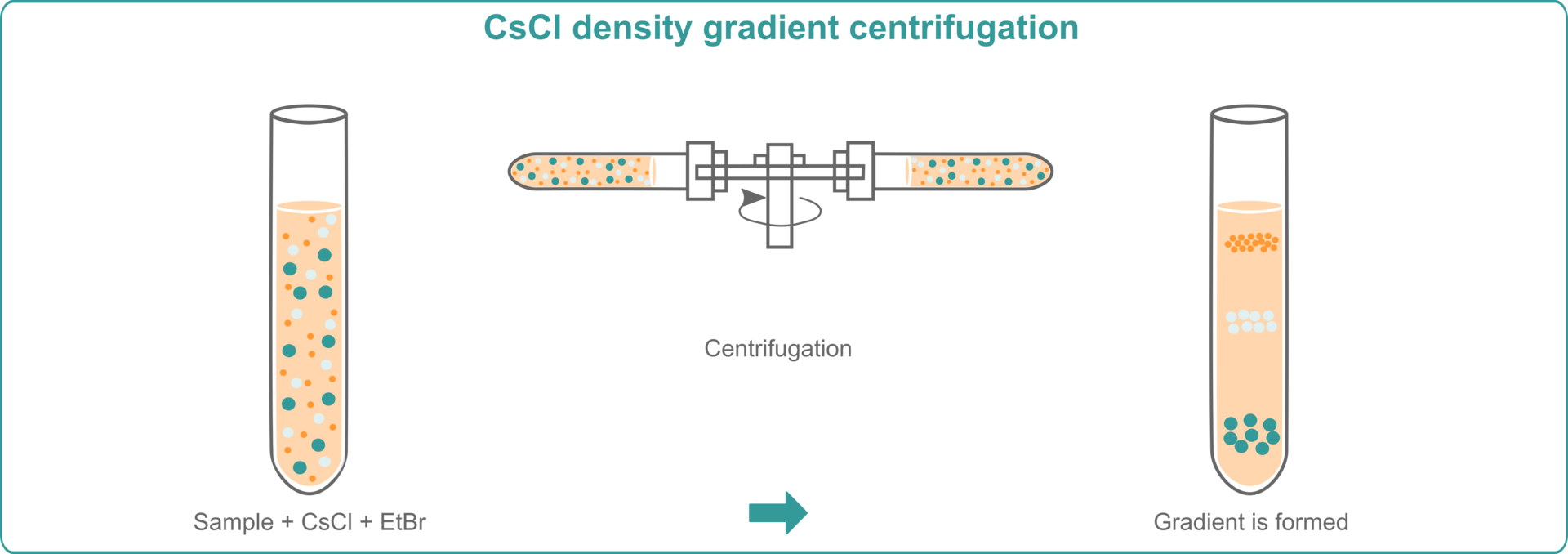
The advantage of this method is that it provides good yields of high purity nucleic acids. However, the disadvantages are that it's laborious and time consuming – because the samples need to be centrifuged for at least 24 hours – and it’s costly because you need an expensive ultracentrifuge, which is a piece of equipment that you probably would not otherwise need. You also have to be careful when working with EthBr, as it’s a mutagen.3,4,5,6 You will therefore need a biosafety cabinet for the pipetting steps involving EthBr if you're planning to use this nucleic acid extraction method in your lab.
Phenol-chloroform extraction
To perform phenol-chloroform extractions, you need to mix your lysed samples with a phenol-chloroform solution and centrifuge them for a few minutes. After centrifugation, you will see three phases: an upper aqueous phase containing nucleic acids, a lower organic phase of lipids, and an interphase comprising proteins. Remove the aqueous phase and use ethanol precipitation to purify and concentrate the DNA.7,8 If you want to extract RNA, add guanidinium isothiocyanate to the phenol-chloroform mixture before centrifuging your samples. This will split them into an upper phase containing RNA and a lower phase containing DNA and proteins. Add isopropanol to precipitate RNA from the upper phase.9
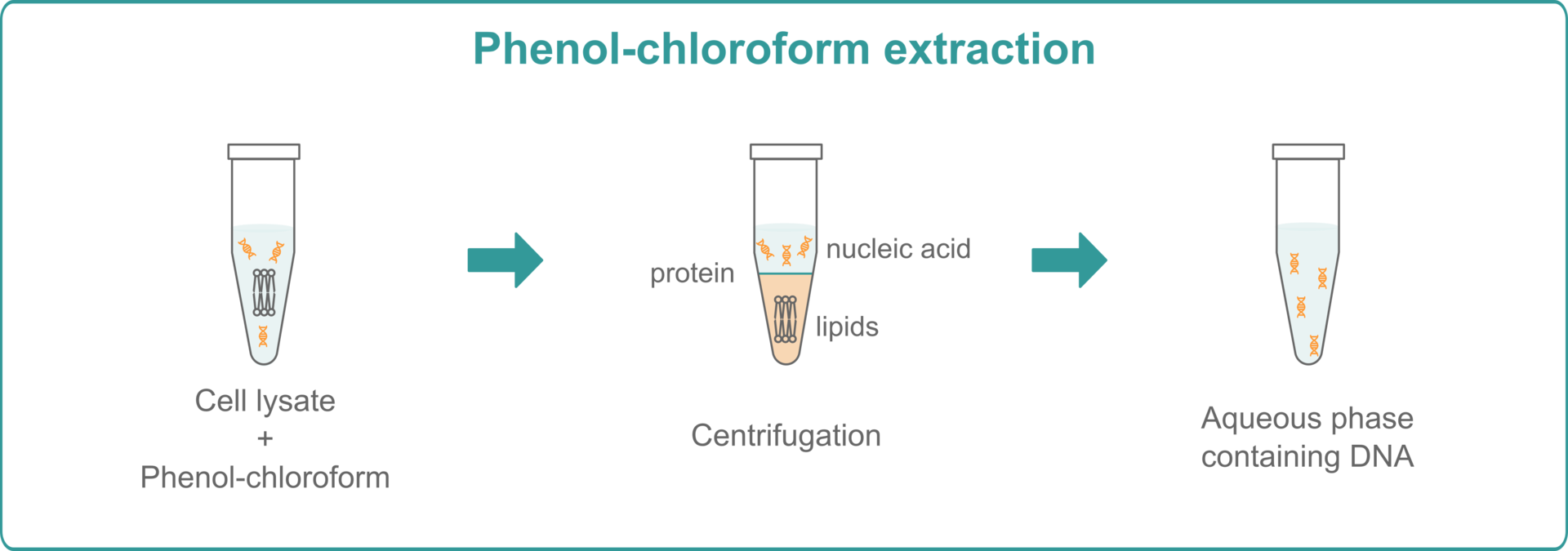
Just like CsCl density gradient centrifugation with EthBr, phenol-chloroform extraction provides high yields and, since you only need a centrifuge instead of an ultracentrifuge, it's much faster and more economical. However, compared to the solid phase extraction methods below, it’s still very time consuming and usually needs to be performed manually, which leads to higher variability and lower reproducibility. In addition, if you want to perform phenol-chloroform extractions, you will not only need to purchase a centrifuge, but also a chemical fume hood, as phenol and chloroform are volatile and highly toxic, and shouldn't be handled on an open bench.
Solid-phase extraction methods
In a nutshell, solid phase extraction can be defined as follows: nucleic acids are bound to a solid surface – such as a silica membrane or magnetic beads – unwanted unbound materials are washed away, and the nucleic acids are later detached from the solid phase. The two most common solid phase extraction methods that we’ll look at in more detail are spin column extraction and magnetic bead extraction.
Spin-column extraction
Spin-column extraction is usually performed with a specific kit. The various kits available on the market differ slightly from each other, but all follow the same basic principle. First, you need to lyse your samples by adding a lysis buffer. Chemical lysis is often combined with physical methods such as bead beating or shaking to break down the cell membrane. Then, the samples are transferred into spin columns. These are centrifuged and nucleic acids bind to the membrane inside the column while other unwanted materials pass through it. To wash away all non-bound components, several centrifugation steps with a wash buffer are needed. At the end, you have to add an elution buffer to the spin columns to liberate the nucleic acids from the membrane and centrifuge them one last time to elute the nucleic acids.
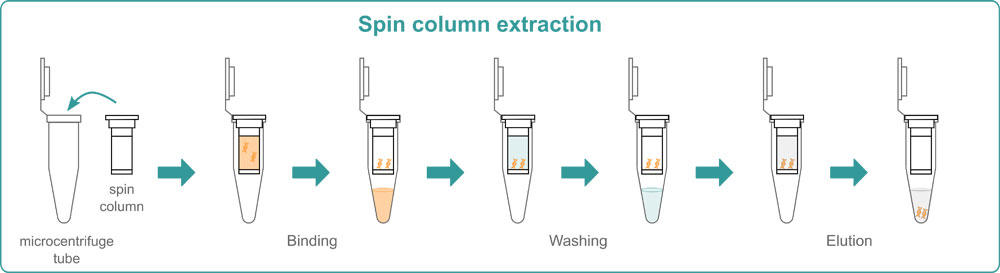
Spin columns are available as individual columns or in a 96 well format. The 96 well silica membrane plates can also be placed on a vacuum manifold instead of being centrifuged. This means that you either need a centrifuge or a vacuum manifold with a pump to perform this method in your lab. The advantages of spin column extraction are that it’s quick and easy to perform, and that you can adapt it to the number of samples that you have. Use single columns if you have few samples, and the 96 well format if you need higher throughput. The major drawbacks are that the membrane can sometimes get clogged, and that a minimum elution volume of 30-50 μl is required, which leads to lower nucleic acid concentrations.
Magnetic bead extraction
Magnetic bead extraction is also performed with specific kits. As with spin column extraction methods, you need a lysis buffer and, depending on the sample, a physical lysis method to disrupt the cells. After sample lysis, magnetic beads that bind the nucleic acids of your samples are added. The tubes are then placed on a magnet, and the supernatant is aspirated to remove unwanted unbound material. This step is repeated several times, replacing the wash buffer in between. For the final stage, an elution buffer is added to detach the nucleic acids from the beads, before transferring the samples to a different vessel.
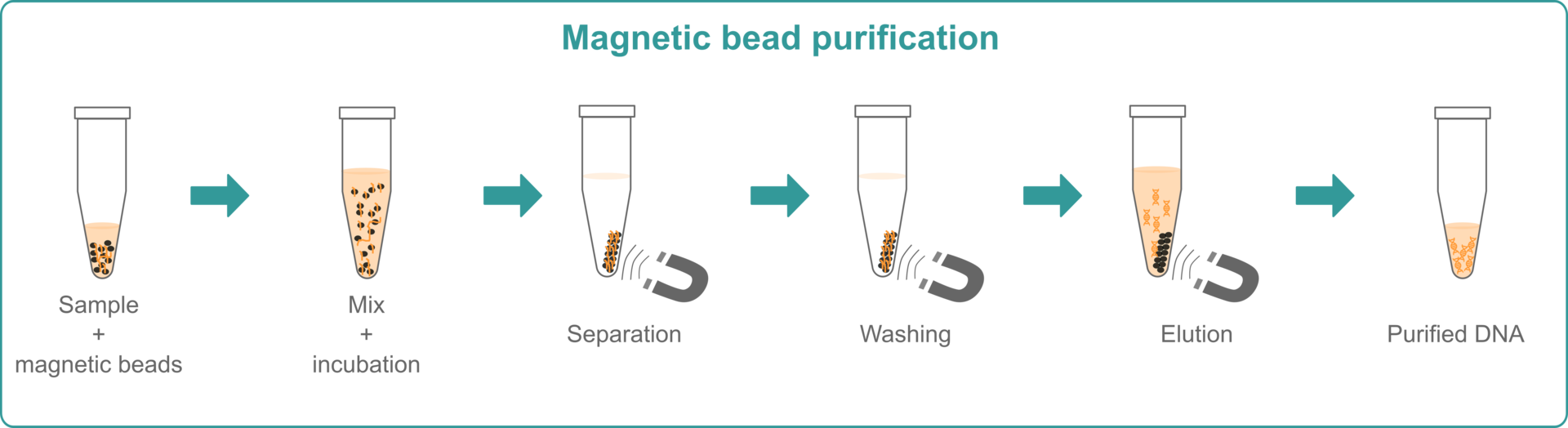
If you wish, you can also watch our video explaining in more detail how the method works.
The huge advantage of this method is that the equipment can be adapted to your budget. Basically, all you need for magnetic bead extraction is a magnetic stand. However, if you don't want to perform the workflow manually, you can also buy a dedicated purification system. It works with magnetic beads too, but typically uses several vessels pre-filled with the different buffers, and then transfers the beads from one vessel to the next using magnetic rods. The third option for those who want to reduce manual pipetting steps, but don't have the budget to get a purification system, is to purchase a benchtop pipetting robot, or a 96 or 384 channel pipette. Both devices reduce manual liquid handling steps and increase throughput, and can also be used for other applications in the lab.
Compared to the spin column method, magnetic bead extraction can work with lower elution volumes, and its throughput can be ramped up more easily, because you can work in a 384 well format. However, manually performing magnetic bead extractions is more tedious and error prone, as you need to be careful not to aspirate the magnetic beads.
Which method did we choose?
As explained in the beginning, we’ve recently set up a new in-house lab and had to decide what type of RNA and DNA extraction method to use and what equipment to purchase.
Ultimately, we decided to go with both spin column and magnetic bead extraction methods, and purchased a centrifuge, vacuum manifold and pump, and magnetic stands. This gives us the flexibility to work with a wide range of kits and meet different throughput requirements. If we have a very low sample number, we can extract nucleic acids quickly by using single spin columns and the centrifuge. If we have higher sample numbers, we can use either 96 well silica membrane plates and the vacuum manifold, or the magnetic bead extraction method.
Conclusion
We hope that our evaluation of the different RNA and DNA extraction methods helps you decide which equipment to choose for your own lab. In the next part of our blog series on how to set up a molecular biology lab from scratch, we’ll focus on what you need to consider when it comes to PCR – check it out to get even more useful tips!



























