-
Master the art of pipetting and learn how to perform serial dilutions with our comprehensive guide
Perform serial dilutions like a pro!
A serial dilution is the step-wise reduction of a concentration using a constant dilution factor and similar volumes. This highly standardized procedure has applications in drug discovery, microbiology and molecular biology. We provide more information on serial dilutions and the related calculations in our detailed blog post on how to do serial dilutions.
We will discuss how to perform serial dilutions with optimized liquid handling parameters to prevent common issues leading to accumulated errors during inter-dependent steps. Half-hearted mixing using insufficient force, slow speeds, or too few mixing steps has the highest impact on the homogeneity of a serial dilution, but you may have questions about how to avoid these problems. For example:
- How do you create the best mixing force to guarantee homogeneity?
- Which pipette is ideal for your dilution?
- Should you change tips between dilution steps?
- Is poor mixing the only issue in your serial dilutions, or can you also improve your results at another point?
We will show data from a water-based serial dilution set-up, isolating key parameters to demonstrate how they influence reliability. The tips provided are a guideline to help you quickly adapt to changing conditions during liquid handling protocols.
Ultimately, you can use our optimized automation protocols for the ASSIST PLUS pipetting robot to eliminate any operator influence. Upscale your knowledge and become a pro at performing serial dilutions with INTEGRA's versatile electronic pipettes on the ASSIST PLUS!
-
Table of contents
A serial dilution is the step-wise reduction of a concentration using a constant dilution factor and similar volumes. This highly standardized procedure has applications in drug discovery, microbiology and molecular biology. We provide more information on serial dilutions and the related calculations in our detailed blog post on how to do serial dilutions.
We will discuss how to perform serial dilutions with optimized liquid handling parameters to prevent common issues leading to accumulated errors during inter-dependent steps. Half-hearted mixing using insufficient force, slow speeds, or too few mixing steps has the highest impact on the homogeneity of a serial dilution, but you may have questions about how to avoid these problems. For example:
- How do you create the best mixing force to guarantee homogeneity?
- Which pipette is ideal for your dilution?
- Should you change tips between dilution steps?
- Is poor mixing the only issue in your serial dilutions, or can you also improve your results at another point?
We will show data from a water-based serial dilution set-up, isolating key parameters to demonstrate how they influence reliability. The tips provided are a guideline to help you quickly adapt to changing conditions during liquid handling protocols.
Ultimately, you can use our optimized automation protocols for the ASSIST PLUS pipetting robot to eliminate any operator influence. Upscale your knowledge and become a pro at performing serial dilutions with INTEGRA's versatile electronic pipettes on the ASSIST PLUS!
Downloads: How to perform serial dilutions - a comprehensive guide
What is the purpose of serial dilutions?
The process of decreasing the concentration of a substance with a diluent (e.g. water) is called dilution, and this is a highly standardized technique. Depending on the desired concentration, dilutions are performed directly or within a series of equal steps (Figure 1). The main advantage of a serial dilution over a direct dilution is the ability to achieve a similar change in concentration with higher transfer volumes.
One purpose of serial dilutions is to create a calibration curve necessary for any application requiring the quantification of an unknown concentration. A calibration curve uses a set of standardized known analytes – covering a broad concentration range – and represents the changing value of a measurable property of a liquid with respect to its concentration. An example of an application that relies on a calibration curve is the enzyme-linked immunosorbent assay (ELISA). You can find even more information about this technique in our detailed blog post about ELISA.
The relationship between concentration and absorbance
A calibration curve usually represents the relationship between concentration and absorbance measurements, but other comparable properties include fluorescence, cell count, PCR product, etc. As soon as the relationship between 2 quantities is directly proportional, it is possible to create a calibration curve by measuring twice at different concentrations (orange). Linear regression analysis (green) defines the relationship between variables, where y represents the measurement value, with x as its concentration. The calibration curve's equation uses the y-value to calculate the x-value.
Serial dilutions also offer benefits to screening-related applications. The detection of minimal concentrations close to the limit of detection (LOD) is only possible when sequentially diluting a known concentration with a fixed factor. For example, in microbiology, serial dilutions help determine the minimum inhibitory concentration (MIC). Our comprehensive blog post about MIC provides all the information you need to perform serial dilutions for this specific application. We also offer protocols for automated MIC testing on the ASSIST PLUS pipetting robot.
How to perform serial dilutions with high accuracy
Serial dilutions crucially depend on excellent liquid handling. The following chapters will provide tips on minimizing errors to improve the reproducibility and reliability of your dilution results. You will learn about essential key parameters in serial dilutions and what happens when these parameters are incorrect!
How inaccurate is a serial dilution when performed incorrectly?

Choosing the right INTEGRA pipette for serial dilutions
When choosing the best pipette, the transfer volumes should be within the range of 35 to 100 % of the nominal volume of the pipette, with consideration that at least 50 % of the total volume is mixed (>80 % of the whole GRIPTIPS® pipette tip volume). Performing a 2-fold serial dilution with a similar transfer and mixing volume is easily possible with our EVOLVE well-balanced manual pipette, VIAFLO lightweight electronic pipette or VOYAGER adjustable tip spacing pipette (Table 1).
Benefit from dynamic volume adjustment when performing 5- and 10-fold serial dilutions with INTEGRA's VIAFLO electronic pipette and VOYAGER adjustable tip spacing pipette.
Using only a single pipette might not be ideal when performing 5- and 10-fold serial dilutions with small transfer volumes. It is crucial to consider which factor will cause the most inaccuracy – going below 35 % of the nominal volume or mixing with much less than 50 % of the total volume. INTEGRA pipettes are calibrated at 10 % of the nominal volume, yet the mixing volume would still be below 50 %. Using different pipettes between the transfer and mixing steps is nothing but impractical. Luckily, with INTEGRA's electronic pipettes, you can benefit from dynamic volume adjustment (Table 2).
Interested in how to use a pipette correctly?
We provide you with 10 important tips in our detailed blog post.
Manual versus automated pipetting
The ASSIST PLUS offers optimized parameters to guarantee effortless aqueous serial dilutions, providing reproducible results and preventing error accumulation:
- Transfer volume > 10 % of nominal pipette volume
- Prewetting of the whole GRIPTIPS 3 times
- 5-10 % pre- and post-dispense during diluent distribution
- 5 times mixing at speed 10 with ~80 % volume and fixed height (lower half of the well)
- Blowout after mixing, no tip change
We diluted 0.36 mM of a tartrazine/water mixture into 96 well flat-bottom plates and analyzed the performance of serial dilutions with a plate reader (Tecan Infinite® M200 PRO) at 428 nm absorbance. The procedure was performed manually with a mechanical pipette and using the ASSIST PLUS pipetting robot with a VOYAGER adjustable tip spacing pipette and optimized settings.
We analyzed a 10-step, 2-fold serial dilution and a 4-step, 5-fold serial dilution at the LOD in 3 independent approaches (n=3). The accuracy and precision of single dilution steps were calculated, with consideration for the target concentration. By changing one parameter at a time, we analyzed the influence of parameters on diluent distribution and serial dilution by comparing the manual serial dilution with the ideal experimental set-up on the ASSIST PLUS.
Not sure how to calculate accuracy and precision?
Take a look at our detailed data analysis guidelines.
The procedure for how to perform serial dilutions (e.g. 5-fold) is illustrated in Figure 2 and divided into the following steps:
- Distribution of 80 µl of diluent (water) to the 96 well flat-bottom plates
- Transfer of 20 µl of highly concentrated tartrazine into the first column
- Mixing
- Transfer of 20 µl of 5-fold diluted tartrazine into the second column
- Repeat
One column contained only the diluent, functioning as a blank to eliminate background absorbance.
Changes to the angle and plunger movement of the mechanical pipette during manual 2-fold serial dilutions (Figure 3a) and 5-fold serial dilutions (Figure 3b) resulted in a user-caused accumulated error of up to 9.5 % inaccuracy. Especially during the 5-fold serial dilution, the transfer volume of 20 µl was insufficient during mixing, resulting in faster error accumulation within fewer dilution steps. Furthermore, both manual serial dilutions showed less reproducibility at the LOD.
In contrast, automation on the ASSIST PLUS provides reproducible results and consistent accuracy of more than 99 % (Figure 3).
Mixing
Optimal mixing of watery liquids is carried out at speed 10 for 5 cycles with ~80 % volume.
Automating serial dilution protocols on the ASSIST PLUS can eliminate user influence to enhance reproducibility, but setting the correct mixing parameters is crucial, as poor mixing can result in inaccuracies of up to 24.5 %.
Yet, what defines good mixing?
When transferring an analyte into the diluent, the analyte is typically in a heterogeneous, clumped state. After thorough mixing, the analyte is evenly distributed in a homogeneous solution (Figure 4).
We analyzed 5 parameters during mixing:
- Speed
- Cycle
- Volume
- Tip travel
- Tip clearance
During the 2-fold serial dilution (Figure 5a) and 5-fold serial dilution (Figure 5b), inaccuracy was observed when decreasing the number of mixing cycles from 5 to 3, or reducing the mixing speed from 10 to 5, with mixing speed having the most impact on accuracy. Mixing only with the transfer volume of 20 µl during the 5-fold serial dilution also caused error accumulation and inaccuracy (Figure 5b). In addition, a combination of errors in the various parameters – such as mixing too slowly (speed 5), using too few mixing steps (3 cycles), and pipetting lower volumes (20 µl) – led to dramatic inaccuracy and error accumulation during the 5-fold serial dilution (Figure 5c).
Do not fail your serial dilutions! Keep the mixing speed at level 10, use 5 mixing cycles, and use ~80 % of the GRIPTIPS volume to minimize inaccuracy during 2-fold (Figure 5a) and 5-fold (Figure 5b, 5c) serial dilutions of aqueous solutions.
But how do you perform serial dilutions when the liquids used do not allow rapid mixing?
If a liquid has poor solubility and requires a reduced mixing speed, ramping up the number of mixing cycles can help to maintain accuracy.
Even aqueous solutions change their characteristics with different analytes, and may not mix rapidly, forcing you to slow down during mixing. During the 2-fold serial dilution (Figure 6a) and 5-fold serial dilution (Figure 6b), slow mixing at speed 5 – combined with only 3 mixing cycles – led to high inaccuracy, error accumulation and reduced precision.
When mixing slowly, consider ramping up the number of mixing cycles to 10 for 2-fold serial dilutions (Figure 6a), or even higher for 5-fold serial dilutions (Figure 6b), to prevent inaccuracy.
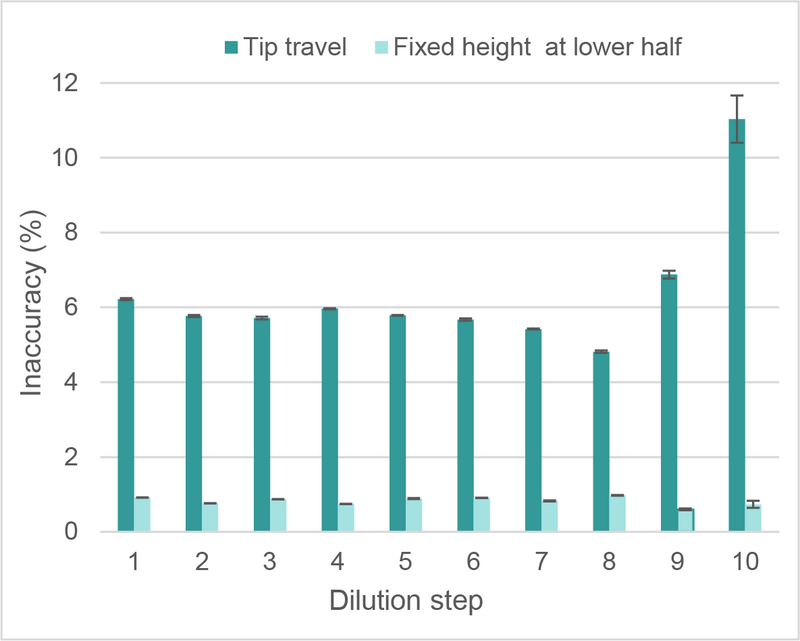
Speaking about dilution-factor independent mixing parameters, what about tip travel during mixing?
Clever mixing avoids tip travel when performing a serial dilution of aqueous solutions.
To test the influence of tip travel (z-travel of GRIPTIPS) during mixing, we performed a 2-fold serial dilution with 2 ml volume in deep well plates, using the 8 channel 1250 µl VOYAGER on the ASSIST PLUS. Using the tip travel function during mixing led to consistent inaccuracies with slight error accumulation in the last 2 dilution steps (Figure 7).
Deselecting the tip travel function, and setting a fixed height at the lower half of the well, helps to produce a stronger mixing force and minimizes inaccuracy (Figure 7).
Finally, should tips be changed during serial dilution steps?
There is no need to change GRIPTIPS during serial dilutions of aqueous solutions, as long as a blowout is performed after each mixing step to prevent liquid carryover.
Performing a tip change after each mixing step without prewetting the whole GRIPTIPS 3 times led to inaccuracy, error accumulation and less precision during a 2-fold serial dilution (Figure 8). Similarly, continuously using the same GRIPTIPS pipette tip during a serial dilution without performing a blowout to completely clear GRIPTIPS of liquid showed inaccuracy but less error accumulation (Figure 8).
For aqueous solutions, you can easily use the same GRIPTIPS during a serial dilution. The blowout after each mixing step will prevent any inaccuracies caused by liquid carryover (Figure 8).
Diluent distribution
Optimal diluent distribution is guaranteed when using 3 times prewetted GRIPTIPS and a 5-10 % pre- and post-dispense.
Another issue causing inaccuracies in serial dilutions happens during diluent distribution. Although automating serial dilution protocols on the ASSIST PLUS eliminates user influence, not prewetting GRIPTIPS or performing pre-and post-dispenses when transferring the diluent causes inaccuracies of ~6 %.
The fastest way to fill a plate with diluent is to use an electronic pipette in repeat dispense mode. Distributing the diluent with 5-10 % pre- and post-dispense and using new GRIPTIPS for every dispense, but not performing a prewetting step, leads to constant high inaccuracy. Similarly, using the same prewetted GRIPTIPS but not performing a pre- and post-dispense causes
overall inaccuracy (Figure 9).
Be smart, and don’t compromise your work with an inaccurate plate set-up. Prewet your GRIPTIPS and perform a 5-10 % pre- and post-dispense to prevent errors caused by erroneous diluent distribution (Figure 9).
Conclusion
- Efficient mixing during serial dilutions combines a fast mixing speed (10) with enough cycles (3) at 80 % of the GRIPTIPS volume or total volume. Additionally, mixing with a fixed height in the lower half of the well creates a greater mixing force.
- If a liquid has poor solubility, ramping up the number of mixing cycles can compensate for slow mixing speeds and help to maintain accuracy.
- Optimal diluent distribution uses 3 times prewetted GRIPTIPS and a 5-10 % pre-/post-dispense.
- Boost your serial dilution's reproducibility and reliability when using the VIAFLO lightweight electronic pipette and VOYAGER adjustable tip spacing pipette with dynamic volume adjustment on the ASSIST PLUS pipetting robot.
- Reward yourself with additional hands-free time when using INTEGRA's electronic pipettes on the ASSIST PLUS.
- Enjoy risk-free processing when working with dangerous solutions, as the ASSIST PLUS fits easily into biosafety cabinets.
Ask our expert. Leave a comment!
Write us if you have any questions regarding the application note or one of our instruments.
Any questions? I'm happy to help!

Download
Download App Note as PDF
DownloadMaster the art of pipetting and learn how to perform serial dilutions with our comprehensive guideCustomer’s voice
We have many operations that are performed in column or plate format and the ASSIST PLUS has drastically reduced the time needed for many of the pipetting steps. As important, they also reduce the risk of error when pipetting many samples.