-
Automated affinity purification of immunoglobulins (IgGs) using INtip™ dispersive solid-phase extraction technology
Purification of antibodies using IMCStips together with the ASSIST PLUS pipetting robot
Solid-phase affinity extraction is a common laboratory method that is widely used in proteomics for protein purification from crude samples. Protein A immobilized resins designed to purify immunoglobulins (IgGs) are available in multiple formats, including magnetic beads, spin columns and 96 well filter plates. Resin-filled IMCStips® offer an alternative approach, employing patented dispersive solidphase extraction (dSPE) technology in a pipette tip (INtip purification) to allow multiple samples to be purified in parallel using multichannel pipettes. The protein purification protocol consists of multiple low speed pipetting cycles and washing steps. Automated liquid handling instruments are the ideal solution to relieve users of the strain caused by long manual pipetting sessions.
INTEGRA’s ASSIST PLUS pipetting robot is designed to automate any of the VIAFLO and VOYAGER multichannel electronic pipettes, freeing scientists from time-consuming, repetitive pipetting tasks for optimum ergonomics.
The integration of INtip purification with INTEGRA’s ASSIST PLUS pipetting robot and VIAFLO 12 channel electronic pipette enables scientists to rapidly and easily automate immunoglobulin purification in their laboratories. The affinity extraction protocol described in this application note demonstrates high recoveries, low background and reproducible antibody enrichment. IMCStips come with predefined pipetting protocols that are uploaded to the VIAFLO electronic pipette using INTEGRA’s VIALINK software.
The ASSIST PLUS pipetting robot is then ready to perform the automated purification protocol, which takes around one hour. This combination of IMCS and INTEGRA Biosciences technologies offers scientists a fast, streamlined workflow that simplifies sample preparation and increases testing efficiency.
-
Table of contents
Solid-phase affinity extraction is a common laboratory method that is widely used in proteomics for protein purification from crude samples. Protein A immobilized resins designed to purify immunoglobulins (IgGs) are available in multiple formats, including magnetic beads, spin columns and 96 well filter plates. Resin-filled IMCStips® offer an alternative approach, employing patented dispersive solidphase extraction (dSPE) technology in a pipette tip (INtip purification) to allow multiple samples to be purified in parallel using multichannel pipettes. The protein purification protocol consists of multiple low speed pipetting cycles and washing steps. Automated liquid handling instruments are the ideal solution to relieve users of the strain caused by long manual pipetting sessions.
INTEGRA’s ASSIST PLUS pipetting robot is designed to automate any of the VIAFLO and VOYAGER multichannel electronic pipettes, freeing scientists from time-consuming, repetitive pipetting tasks for optimum ergonomics.
The integration of INtip purification with INTEGRA’s ASSIST PLUS pipetting robot and VIAFLO 12 channel electronic pipette enables scientists to rapidly and easily automate immunoglobulin purification in their laboratories. The affinity extraction protocol described in this application note demonstrates high recoveries, low background and reproducible antibody enrichment. IMCStips come with predefined pipetting protocols that are uploaded to the VIAFLO electronic pipette using INTEGRA’s VIALINK software.
The ASSIST PLUS pipetting robot is then ready to perform the automated purification protocol, which takes around one hour. This combination of IMCS and INTEGRA Biosciences technologies offers scientists a fast, streamlined workflow that simplifies sample preparation and increases testing efficiency.
Key benefits
- The IMCStips patented dSPE technology is designed to provide simplified and highly efficient sample preparation in proteomics.
- The ASSIST PLUS pipetting robot, together with the VIAFLO electronic pipette, allows full automation of the affinity purification protocols, offering high reproducibility, error-free pipetting, reduced hands-on time and unmatched ergonomics.
- The IgG purification protocol presented in this application note has been optimized to provide consistent results, high recoveries and faster workflows.
- By combining their technologies, IMCS and INTEGRA Biosciences offer scientists a simple, ready-to-use, flexible automated solution for parallel purification of up to twelve immunoglobulin samples.
Experimental set-up
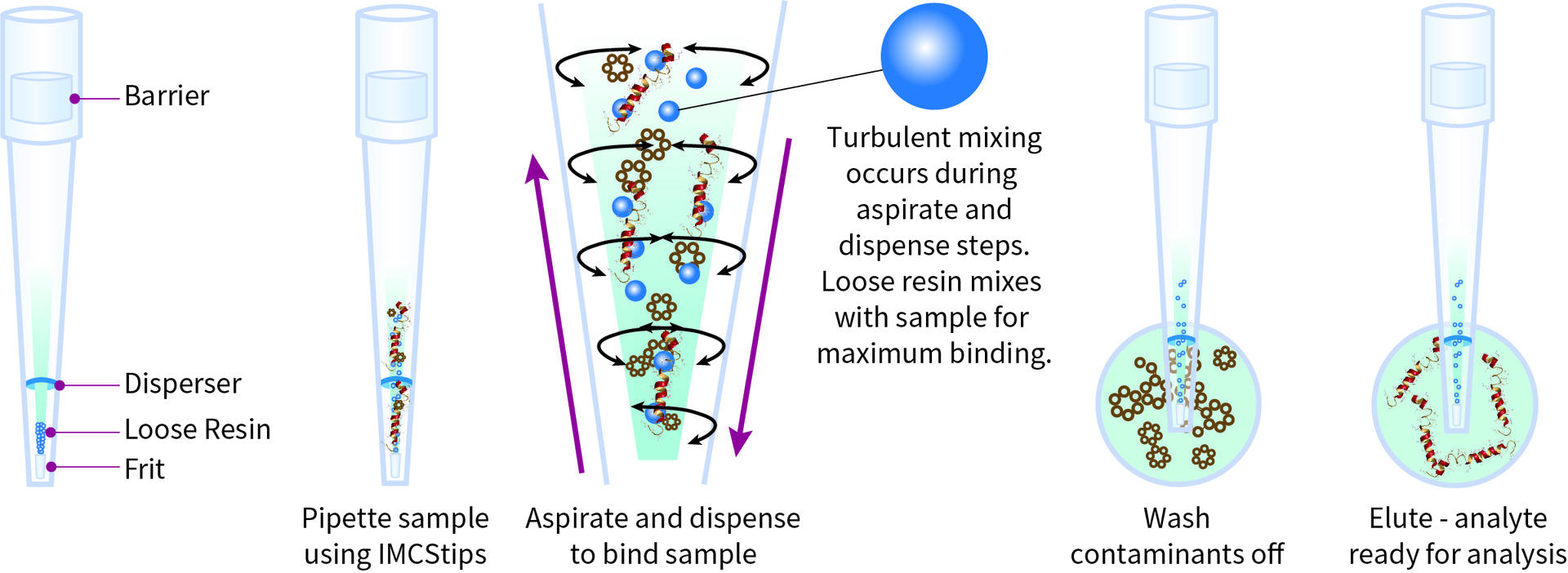
Protein A 1250 μl IMCStips contain loose resin (MabSelect SuRe™ LX) placed between a top and a bottom porous filter. Each tip features a disperser ring located inside the tip that constricts the liquid flow to introduce turbulent mixing between the liquid sample and the resin (Figure 1). The following procedure describes the purification protocol developed and optimized by IMCS together with INTEGRA Biosciences. The suggested extraction buffers have been tested to achieve the best results. The extraction follows a typical bind, wash and elute workflow. The entire IgG extraction process is automated using the ASSIST PLUS pipetting robot together with a VIAFLO 12 channel 1250 μl electronic pipette and 12 Protein A IMCStips. The binding capacity per tip is 1 mg IgG, determined based on more than 80 % recovery of human polyclonal antibody. All other liquid handling steps are performed using 1250 μl Sterile, Filter, Low Retention GRIPTIPS®. The entire protocol is driven by a main program coupled to six sub-methods. The primary program and all six sub-methods are necessary to run the automated purification process on the ASSIST PLUS pipetting robot. INTEGRA’s VIALINK software allows transfer of all the files to the VIAFLO electronic pipette.
Overview of the pipetting steps and corresponding programs:
Main program: primary program that drives the full purification protocol.
Associate sub-programs:
- Reagents (R): distribution of the reagents into the elution plate
- Conditioning (C): preparation of the IMCStips before sample handling
- Samples (S): sample binding onto the resin contained in the IMCStips
- Washing (W): bound sample washing for removing of non-specific binding
- Elution (E): sample elution from the resin
- Neutralizing (N): neutralization of the elution buffer
Notes:
- The program names used in this application note are given to provide an understanding of the purification steps and differ from the delivered programs.
- This application note describes the protocol for the ASSIST PLUS and VIAFLO 12 channel electronic pipette. The data shown was acquired by IMCS using an ASSIST pipetting robot with a VIAFLO 8 channel pipette.

Downloads
1. Pipette set-up
Transfer all VIALINK programs onto the VIAFLO 12 channel 1250 μl electronic pipette and adjust the pipette flow rates.
The IMCStips are delivered with a zip file containing all necessary VIALINK programs. After extracting the files, transfer them onto the pipette using the VIALINK software and the charging/communication stand for one pipette. The pipetting speeds have been specifically optimized for this extraction protocol and differ from the factory-set speeds. Go to the speed table of the VIAFLO pipette, select Toolbox then Pipetting Preferences, and customize the speed according to IMCS protocol recommendations. The VIAFLO electronic pipette is ready to operate.
Tips:
- The VIALINK program used for this protocol is a ready-to-use solution. It combines ease of use, full automation and customization of all necessary protocol settings, freeing scientists from all programming needs or manual interventions.
- Loading the VIALINK programs on the VIAFLO electronic pipette is done in seconds. The charging/communication stand for one pipette allows not only enables connection of the pipette to the INTEGRA software, but also charging and storage between uses.
- INTEGRA’s electronic pipettes come with 10 default pipetting speeds. The speed table on the pipette provides a direct correlation of the speed value to a flow rate expressed in μl/s. The pipetting speed can be easily customized by entering a new flow rate for a speed value. If necessary, dedicate the customized pipette to your specific application, as the speed changes affect all pipetting programs on the pipette.
2. ASSIST PLUS deck set-up
Prepare the ASSIST PLUS deck with the IMCStips, GRIPTIPS, samples and all the reagents required for the purification protocol.
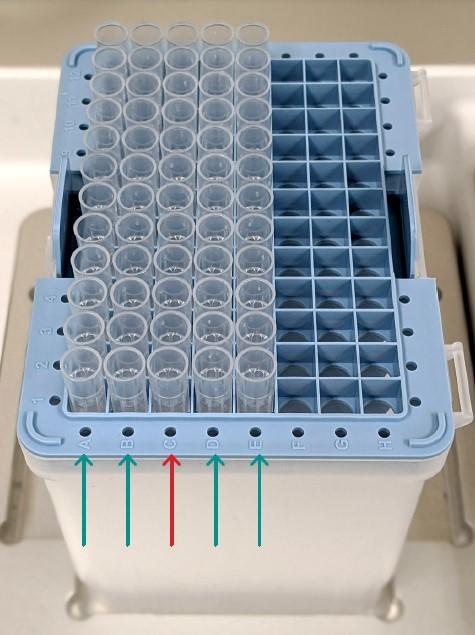
Replace the 12 GRIPTIPS in row C of a 1250 μl Sterile, Filter, Low Retention GRIPTIP rack with 12 Protein A IMCStips (Figure 2).
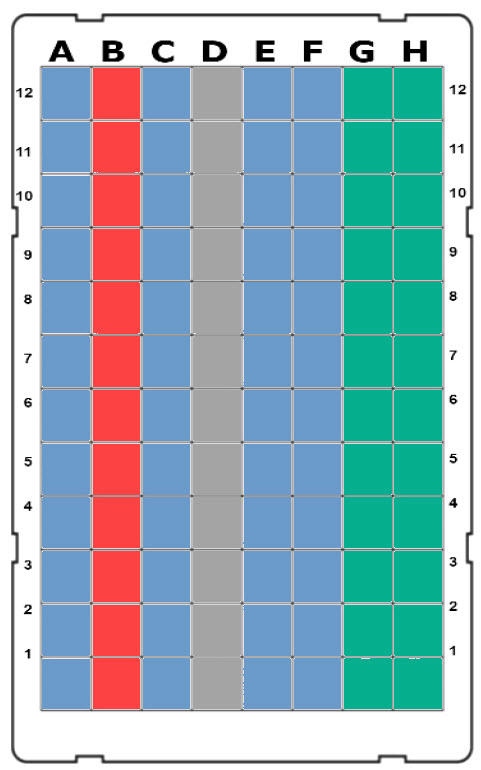
Place the pipette tip box on the ASSIST PLUS. Fill a 100 ml multichannel reagent reservoir with 1x PBS. Prepare the 8 row reagent reservoir with the different reagents as shown in Figure 3. Use a 96 well, deep well plate as the elution plate and manually fill row B with the IgG samples to be purified.
Place the labware with the samples and buffers on the deck of the pipetting robot as indicated in Figure 4.
Tips:
- The IMCStips are supplied with the resin suspended in storage buffer. Before use, remove the silicone cap (top) and the silicone plug (bottom), and use a clean wipe to dry off the liquid flowing out of the tip.


Figure 4: ASSIST PLUS deck set-up. 1: GRIPTIP rack with 12 IMCStips placed in row C. 2 (Position A): 100 ml multichannel reagent reservoir containing 1x PBS. 3 (Position B): 8 row reagent reservoir containing various buffers as shown in Figure 3. 4 (Position C): 96 well, deep well elution plate containing the samples to be purified in row B.
3. Automated elution plate preparation
Transfer the different buffers into the elution plate.
Mount the VIAFLO 12 channel 1250 μl electronic pipette onto the ASSIST PLUS. Select and run the main program, stored on the pipette under the ASSIST PLUS Custom Programs menu. The different reagents are transferred into the elution plate (see Figure 5). Simply set or confirm the loading of the pipette tips from row A to start the program.
4. IMCStips conditioning
Prepare the IMCStips for the sample purification. Equilibrate the resin.
The ASSIST PLUS pipetting robot loads the IMCStips, which are conditioned by aspirating and dispensing 800 μl 1x PBS for three cycles. This step removes all traces of the storage buffer and equilibrates the resin. The IMCStips are ready to purify the IgG samples.
Tips:
- The aspiration and dispense speeds have been optimized to allow the loose resin to be perfectly mixed with the equilibration buffer in the IMCStips. Delay times have been included to allow time for the liquid to pass through the bottom frit of the IMCStips while preserving the binding capacity of the resin.
5. Sample binding
Bind the IgG samples onto the protein A resin.
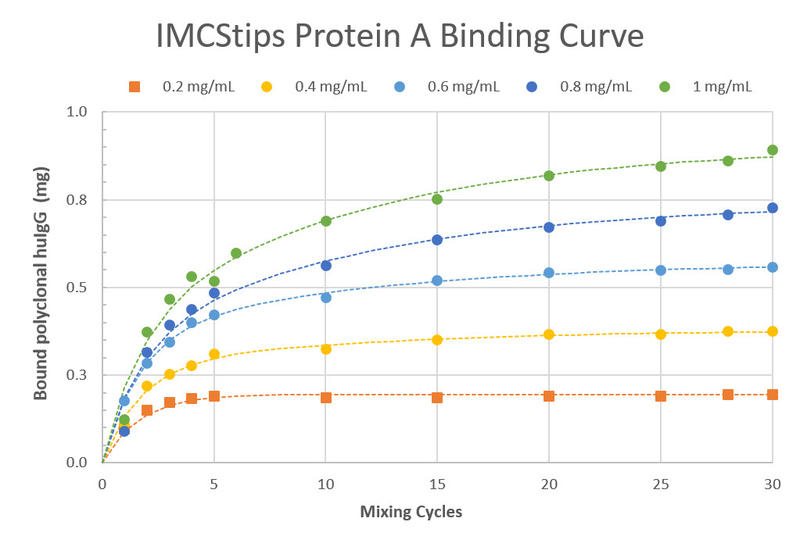
The ASSIST PLUS pipetting robot aspirates 800 μl of each sample and mixes it with the resin for 25 cycles.
The developed method showed experimental results with excellent binding capacity (IMCS, Figure 6).
Tips:
- The IMCStips patented disperser enhances turbulent mixing of the loose resin with the samples for increased binding (Figure 1).
- The mixing cycles – including liquid volumes, delay times, aspiration and dispense speeds, pipetting heights and cycle repetition – have been optimized to ensure maximum contact between the resin and the analytes of interest, and maximum binding rate.
6. Sample washing
Wash the bound samples to remove non-specific binding components and contaminants.
The pipetting robot performs the washing step for the IgG samples bound to the resin (Figure 1). This step includes four consecutive washes with three mixing cycles for each, with the robot moving between washing rows C, D, E and F one after the other (Figure 5).
Tips:
- Buffers, volumes, pipetting speeds and delays have been optimized to ensure consistent and efficient washing to eliminate contaminants.
7. Sample elution
Unbind the purified samples from the resin for further analysis.
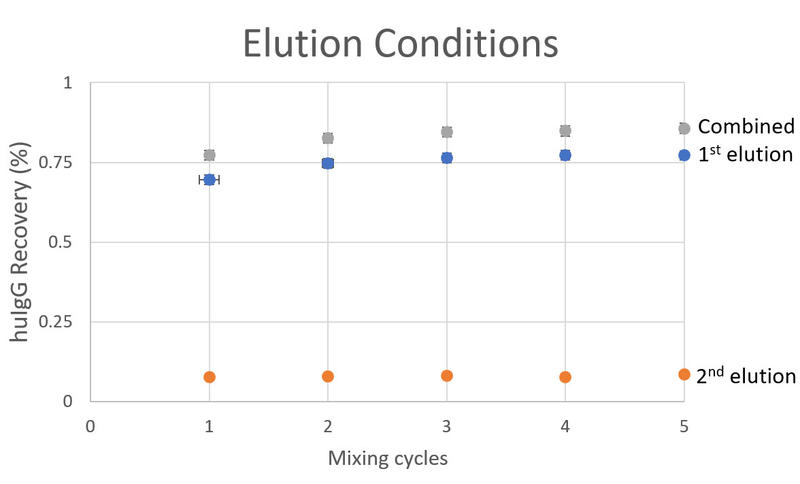
The pipette moves to the first elution row (row G – Figure 5). After five mixing cycles, the ASSIST PLUS transports the IMCStips to the second elution row (row H – Figure 5) for five mixing cycles (Figure 1). Experimental results (IMCS) showed excellent analyte recoveries when using the developed elution method (Figure 7).
Figure 7: The number of mixing cycles is plotted against analyte recoveries. Three mixing cycles were required to achieve consistent recoveries during the first elution step. Due to the dead volume created by the resin and filter, the second elution step provides an additional 10 % analyte recovery. Performing more than three mixing cycles does not enhance recoveries further. Results were obtained using the ASSIST pipetting robot with a VIAFLO 8 channel 1250 μl electronic pipette and IMCStips.
8. Neutralization
Neutralize the elution buffer.
Neutralization buffer (row B of the 8 row reagent reservoir – Figure 3) is transferred to the elution wells (rows G and H – Figure 5) and mixed three times with the elution buffer containing the purified immunoglobulins G.
Remarks
ASSIST:
This affinity purification protocol is also available on the ASSIST pipetting robot and allows the purification of eight samples in parallel in less than 90 minutes. The combination of IMCStips with INTEGRA’s ASSIST pipetting robot is ideal for semi-automated and small-scale purification of immunoglobulins, offering a space-saving, very affordable and accessible entry point in automating sample preparation.
VIALINK software:
The VIALINK programs can be easily adapted to your specific protocols, for instance, for adjustment of the mixing cycles for proof-of-concept workflows.
Conclusion
- Protein A IMCStips and the ASSIST PLUS pipetting robot, together with the VIAFLO electronic pipette, offer fully automated purification of twelve IgG samples in around one hour.
- Experimental results demonstrate the consistent performance of IMCStips for IgG purification using INTEGRA’s pipetting robots.
- The developed protocol offers a simple, fast, ready-to-use and automated workflow for high recoveries, low background, and reproducible antibody enrichments.
- IMCStips are delivered with predefined pipetting protocols to be uploaded to the VIAFLO electronic pipette using INTEGRA’s VIALINK software. The method retains the flexibility for customization.
- For semi-automated and small-scale purification, the affinity purification protocol is also available for the ASSIST pipetting robot and allows the purification of eight samples in less than 90 minutes.
Ask our expert. Leave a comment!
Write us if you have any questions regarding the application note or one of our instruments.
Any questions? I'm happy to help!
Instruments and accessories
ASSIST PLUS, Pipetting Robot
INTEGRA has developed the ASSIST PLUS pipetting robot to streamline routine pipetting tasks at an affordable price. Using INTEGRA electronic multichannel pipettes, the system:
- automates pipetting tasks,
- eliminates physical strain and
- ensures superior reproducibility and
- error free pipetting.
Part No. 4505

VIAFLO 12 channel, 1250 µl, electronic pipette
The Touch Wheel is a quick and ergonomic way to modify pipetting parameters. Instead of repeatedly pushing buttons or twisting fingers to modify volumes, you simply slide your finger over the wheel.
The full color screen provides full text menus (in multiple languages) and displays pipetting protocols without abbreviations, making VIAFLO pipettes particularly easy to understand and intuitive to use.
Part No. 4634
GRIPTIPS®, 1250 µl, Sterile, Filter, Low Retention (for automation systems)
GRIPTIPS® pipette tips perfectly match the multi-lobe tip fitting, snapping firmly on during loading. This guarantees a perfect seal on every tip, preventing them from loosening, leaking or completely falling off. All tips are precisely aligned horizontally, enabling accurate touch-offs, even when pipetting with 384 tips.
Part No. 6545

8 Row Polypropylene Reservoirs (6372)
The polypropylene design improves the reservoirs chemical compatibility and also allows the reservoirs to be autoclaved.
- Reagent reservoirs for 96 well plates
- Pyramid bottom
- Each row holds 32 ml
Part No. 6372

IMCS, Inc.: 1250 µl IMCStips
MCStips® is a patented solid phase extraction technology used in the field of proteomics. Our dispersive pipette extraction technology contains loose resin, which achieves higher binding capacities and consistent, reproducible results.
Source: Website IMCS
Eppendorf: 96/2000 µl Deepwell Plate
Eppendorf® Deepwell plates, standard, 96 wells blue plate, conical bottom colorless wells, capacity 500 μL
Source: Website Eppendorf

Download App Note as PDF
Download App Note "Automated affinity purification of immunoglobulins (IgGs) using INtip™ dispersive solid-phase extraction technology" as PDF
DownloadAutomated affinity purification of immunoglobulins (IgGs) using INtip™ dispersive solid-phase extraction technologyCustomer’s voice
Easy and flexible! The use of standard pipets, ease of use and compact footprint makes it an ideal system for simple lab automation applications without a lot of automation knowledge.